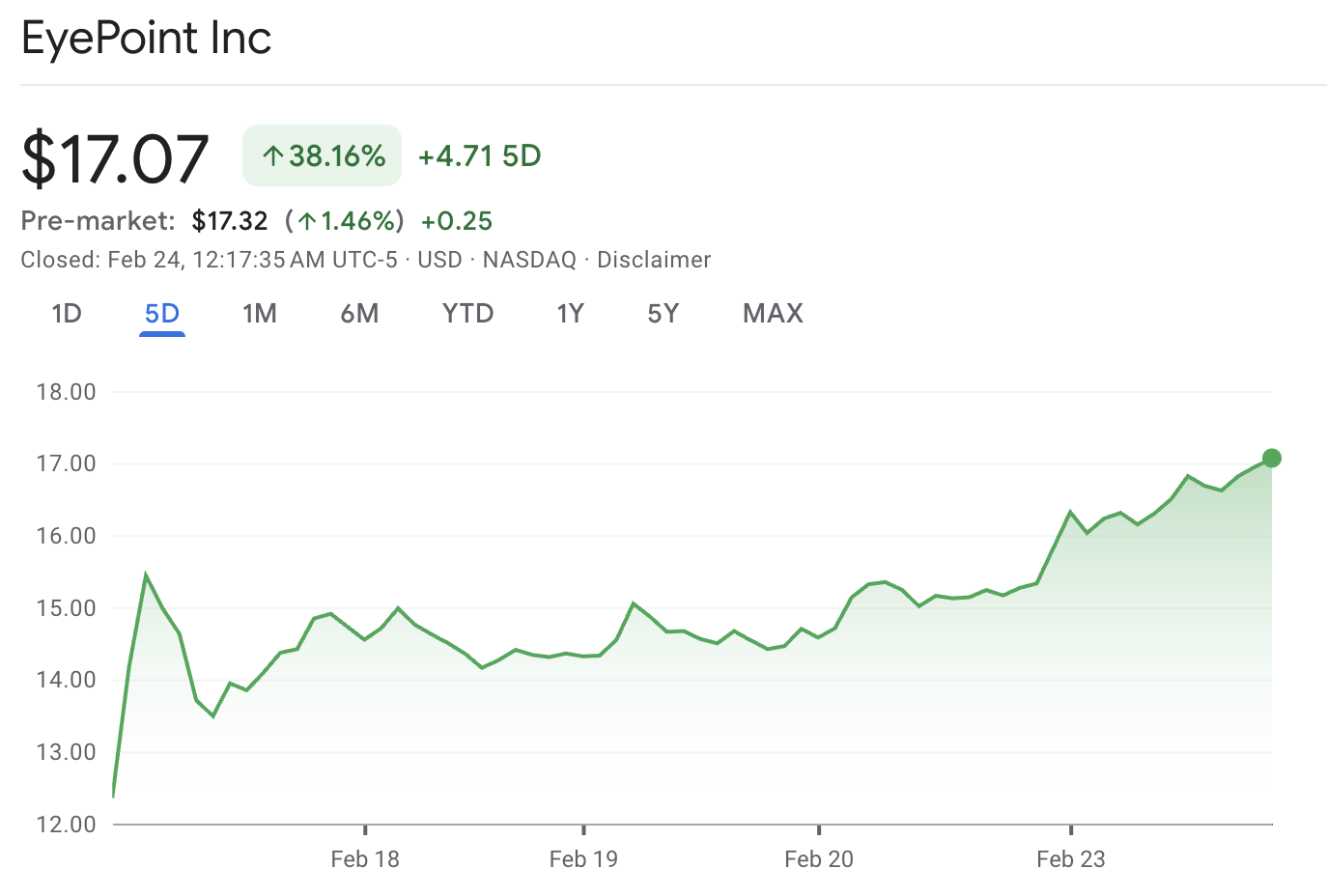
EyePoint (EYPT) is back on traders' screens after a sharp jump in the share price. EYPT stock last traded around $17.07, up about $1.75 on the day, which is roughly an 11% move. It also recorded a wide intraday range, reaching a high of around $17.32 and a low of approximately $14.83, indicating that both buyers and sellers were active.
When a clinical-stage name jumps like this, the market is usually doing two things at once. It is pricing the next catalyst and clearing out positioning that was leaning the wrong way.

In EYPT's case, the catalyst is the anticipated Phase 3 wet AMD data, which are expected to start in mid-2026 with the LUGANO trial, followed shortly by LUCIA.
Thus, should you buy EYPT stock ahead of the Phase 3 wet AMD readout, or is this rally already doing the work that the catalyst would normally do?
Why EYPT Stock Is Surging Now?
The simplest explanation is that the market is moving back toward "catalyst mode." A more thorough explanation consists of three parts.
1) The Phase 3 Calendar Is Getting Closer, and the Setup Is Easy to Understand
EYPT's lead program, DURAVYU (vorolanib intravitreal insert), is currently undergoing two Phase 3 trials for wet AMD, named LUGANO and LUCIA.
EyePoint has announced the completion of patient enrollment for two Phase 3 wet AMD trials, totaling over 900 participants. One company update also specified 432 patients in LUGANO and 475 in LUCIA.
The company expects to release topline data starting in mid-2026.
When a biotech name has a known, high-impact date window, it often attracts:
2) The Company Has Reduced Near-Term Financing Anxiety
EYPT has highlighted approximately $300 million in cash and investments as of December 31, 2025, and said its cash runway extends into Q4 2027.
That's important because the market typically penalizes clinical-stage stocks when it expects an imminent fundraising effort.
3) Short Positioning Can Add Fuel When Price Turns Up
EYPT has also had meaningful short interest. As of January 30, 2026, reports indicated that 8.64 million shares were sold short, representing approximately 10.92% of the public float, with an estimated 9.5 days required to cover.
That does not automatically mean "short squeeze," but it does mean sharp rallies can become sharper if shorts decide to reduce risk at the same time.
The Phase 3 Wet AMD Readout: What Investors Are Actually Betting On
Wet AMD is a leading cause of severe vision loss in older adults. Treatment today often means repeated injections in the eye, which is effective but demanding for patients and clinics.
EyePoint's program aims to address a fundamental question: Can you achieve similar vision outcomes while minimizing the treatment burden?
What the Phase 3 Trials Are Testing
The pivotal program includes two trials: LUGANO and LUCIA. The company has described them as:
Randomized and double-masked
Designed as non-inferiority studies
Comparing DURAVYU (vorolanib intravitreal insert) dosed every six months to an on-label standard-of-care control injection.
Dosing Schedule
Patients in the DURAVYU arm receive a dose every six months, starting at month two of the trial, delivered by an in-office injection.
The Primary Endpoint That Decides the Headline
The primary endpoint is the average change in best-corrected visual acuity (BCVA) at weeks 52 and 56 versus baseline.
Secondary Endpoints That Can Shape the Quality of the Win
Secondary measures include:
Safety
Reduction in treatment burden
Percentage of eyes free of supplemental injections
Anatomical outcomes measured by retinal imaging
In short, even if the trial meets the primary endpoint, the stock reaction can still depend on how clean the secondary story looks.
The Fundamental Reality Check: EYPT Is Still a "Development-Stage" Story
The bullish thesis is about 2026 trial outcomes and what those outcomes unlock. The bearish thesis emphasizes time, cash burn, and the risk that good but not great data could still fall short.
In its Q3 2025 results, the company reported $1.0 million in revenue, $63.0 million in operating expenses, and a net loss of $59.7 million for the quarter.
That kind of cost structure is normal for late-stage development. Still, it reinforces one point: the stock will trade on clinical and regulatory progress more than on current financial performance.
This also explains why the market reacts so strongly to runway comments. If investors believe the company can reach the readout and a filing without a rushed raise, they tend to pay a higher price for the optionality.
So, Should You Buy EYPT Stock Ahead of the Readout?
Buying ahead of EYPT Phase 3 data is not a normal "earnings trade." It is a bet on a medical outcome and the market's reaction to it.
Scenario Table: What Could Happen Next
| Scenario |
What the readout looks like |
What the market often does |
| Bull case |
Meets non-inferiority on BCVA and shows a convincing burden-reduction profile |
Stock can re-rate quickly as investors price a credible approval path |
| Mixed case |
Meets the main endpoint, but rescue injections are higher than hoped or safety is debated |
Stock may spike, then fade as investors argue over “how commercial” it really is |
| Bear case |
Misses non-inferiority or shows an uncomfortable safety signal |
Stock can gap down sharply and stay volatile for weeks |
This is not a prediction. It is a way to understand why "buying ahead" can be profitable but unforgiving.
Risk Checklist Before You Buy Ahead
If you are considering buying after the surge, these questions matter more than price targets:
Can you hold through a large gap move in either direction?
Are you buying for the Phase 3 event, or for the longer-term product story?
Would you prefer to scale in over time rather than buy all at once?
Do you have a clear exit strategy if the stockdrops 20% to 40% quickly?
With biotech, the wrong size is often a bigger problem than the wrong opinion.
Key Catalysts to Watch Next
If you are following EYPT from here, these are the events most likely to move the stock before the Phase 3 readout.
More updates on the mid-2026 data window for LUGANO and LUCIA.
Any safety committee updates?
DME Phase 3 program starts
Short interest changes because a rising days-to-cover number can amplify volatility both up and down.
Frequently Asked Questions
Why Did EYPT Stock Surge?
EYPT's stock surged as the market refocused on the Phase 3 wet AMD data expected in mid-2026, following a series of company updates that reinforced their readiness. This included a new commercial leadership hire and reiterated the trial timelines.
When Is the Phase 3 Wet Amd Readout?
The company has indicated that topline data from the two pivotal trials is expected to be released in mid-2026.
What Does the Phase 3 Program Need to Prove?
The primary endpoint is to demonstrate non-inferiority in the average change in Best Corrected Visual Acuity (BCVA) from baseline at weeks 52 and 56. Secondary measures will focus on safety and treatment burden.
Is EYPT Stock Overbought Right Now?
In the latest daily technical snapshot, the RSI (14) was at 76.165, indicating it is in overbought territory. Being overbought does not mean it "must fall," but it often suggests a higher risk of a short-term pullback.
Conclusion
In conclusion, EYPT is surging because investors are repositioning for a mid-2026 Phase 3 wet AMD readout, and the company has reduced near-term funding pressure with a cash runway it says extends into Q4 2027.
The readout is designed to demonstrate noninferior vision outcomes at weeks 52 and 56, while supporting the argument for fewer treatments, which is where the "real upside narrative" lies.
Buying ahead of Phase 3 data can work, but it is never a free trade. The same catalyst that creates upside can also create a sudden gap down if the data disappoints. If you are considering exposure after a big move, focus on position size, a clear plan, and the technical levels that define whether the trend remains intact.
Disclaimer: This material is for general information purposes only and is not intended as (and should not be considered to be) financial, investment or other advice on which reliance should be placed. No opinion given in the material constitutes a recommendation by EBC or the author that any particular investment, security, transaction or investment strategy is suitable for any specific person.