On March 3, 2026, Moderna agreed to pay $950 million upfront to settle a long-running patent dispute tied to the lipid nanoparticle (LNP) delivery technology used in its COVID-19 vaccines. There is a potential additional payment of $1.3 billion, depending on the outcome of a related legal issue.
The trade-off is clear. The company is choosing certainty over optionality by writing a large check, and it is doing so while investors are still debating how quickly Moderna can rebuild steady revenue beyond its COVID franchise.
That is why the real investment question is not whether the settlement is "good" or "bad." The real question is whether the settlement meaningfully improves Moderna's risk profile enough to justify holding through the next catalyst cycle, or buying into volatility.
Should You Hold or Buy MRNA Stock After the $950M Settlement?

Firstly, the settlement is a good starting point because it removes a major uncertainty about Moderna's vaccine portfolio. However, it also entails a significant cash cost and retains a smaller legal "tail risk" under the contingent payment structure.
In practical terms, the deal can support sentiment by clearing a cloud over current and next-generation infectious-disease products. Nonetheless, MRNA stock now faces more difficult fundamentals than during the pandemic, with reduced COVID-related revenue, ongoing losses, and waning investor patience regarding pipeline timelines.
If You Are Already Holding MRNA Stock, These Are the Hold Signals
You believe the settlement meaningfully reduces long-term uncertainty around respiratory commercialization and next-generation product planning.
You are at ease with an anticipated $950 million cash outflow in Q3 2026, given that Moderna forecasts significant liquidity by the end of the year.
You see upside optionality if the Section 1498 appeal outcome avoids the contingent payment.
If You Are Considering Buying, These Are the Potential Buy Conditions
You want confirmation that fundamentals, not only headline relief, are supporting the settlement-driven rally.
You want visibility on how management will manage cash, pipeline spend, and any additional legal exposures.
You want a cleaner technical setup where the stock regains key moving averages and holds them.
Buy vs Hold Decision Checklist
| Category |
What to check |
| Legal tail risk |
Timeline and probability for the Section 1498 appeal outcome |
| Liquidity |
Management’s cash and liquidity guidance into Q3 2026 |
| Product execution |
Next trial readouts and regulatory milestones |
| Technicals |
Whether price reclaims and holds the key trend levels |
What Happened to Moderna Today?
Moderna has agreed to pay $950 million to settle a major patent dispute related to its lipid nanoparticle (LNP) technology.
The settlement is with Genevant Sciences (a Roivant subsidiary) and Arbutus Biopharma, and it resolves U.S. and international enforcement actions related to Moderna's alleged unauthorized use of LNP technology in its COVID-19 vaccines, including Spikevax.
Based on the public announcement from Roivant (Genevant's parent), the agreement includes:
$950 million upfront to Genevant and Arbutus, with the release stating the payment is expected in July 2026.
A contingent $1.3 billion payment is tied to the outcome of Moderna's Section 1498 appeal-related issue described in the release.
A licensing structure and settlement terms aim to conclude enforcement actions related to the dispute and establish a path forward.
MRNA Stock Initial Reaction
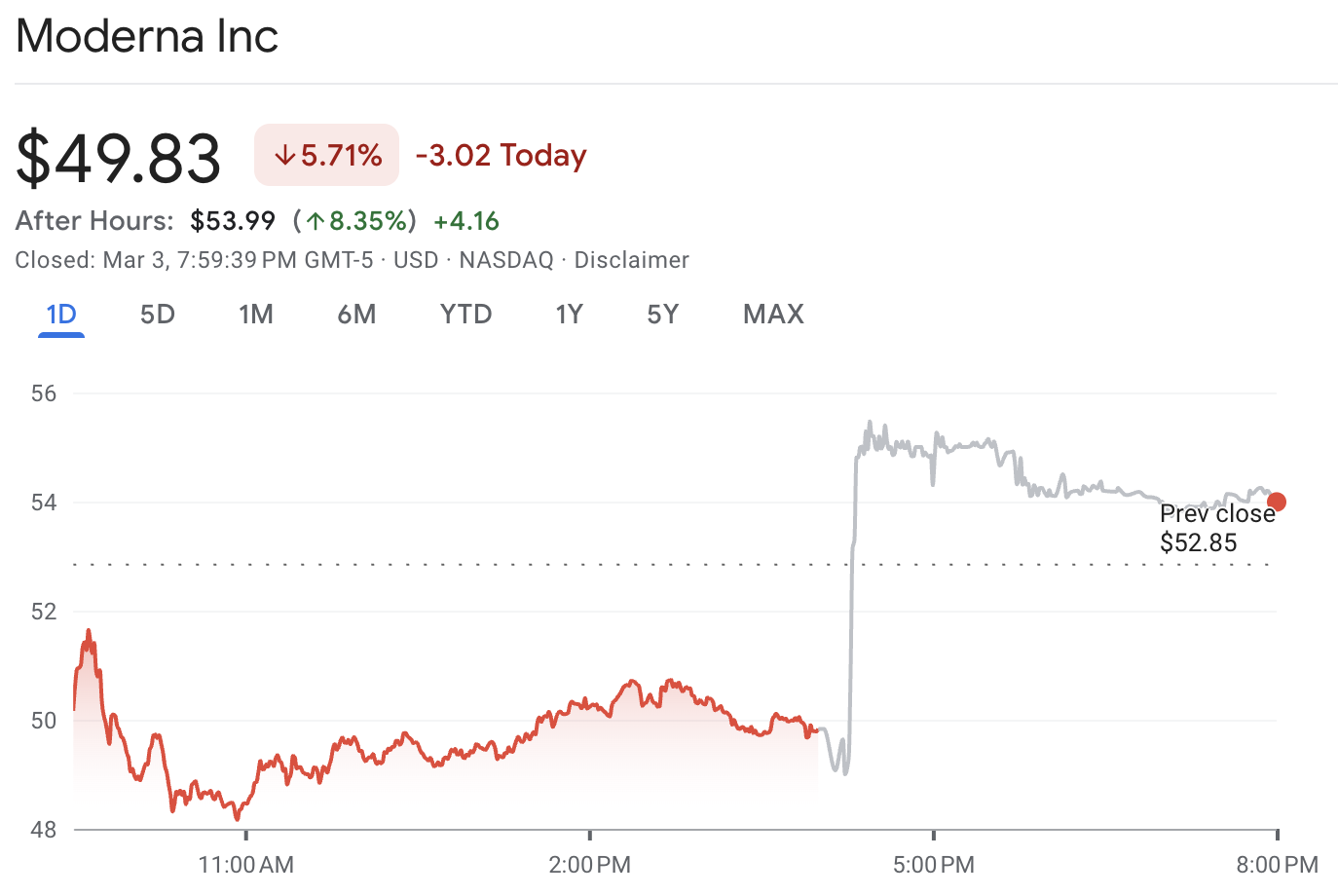
On the day the settlement hit, MRNA stock rose sharply, including an after-hours jump that pushed shares toward the mid-$50s, as the deal "clears" part of the vaccine pipeline uncertainty.
That reaction makes sense for three reasons:
It removes injunction anxiety.
It makes pricing and launch plans easier to model.
It frees management attention.
The Unfortunate Reality: $950M Is Only the First Layer of the Risk
What Is Section 1498 and Why Does It Matter?
Section 1498 is a U.S. statute that can shift patent liability to the government when an invention is used "for the Government," which in practice can limit claims against contractors and direct patent holders toward claims against the U.S. government in the Court of Federal Claims.
In this settlement, the additional $1.3 billion is tied to an appellate ruling on whether Section 1498 bars most of Genevant's and Arbutus's claims against Moderna, except for certain doses characterized as having been delivered to U.S. government employees.
In short, Moderna intends to appeal, claiming it is entitled to government contractor immunity. If the appeal is unsuccessful, Moderna may have to pay an additional $1.3 billion within 90 days.
What the Settlement Changes Mean for Moderna's Product Strategy
The settlement eliminates the need for Moderna to pay future royalties on sales of Spikevax and mResvia. Additionally, it eliminates the uncertainty regarding litigation for next-generation programs such as mNexspike and mCombriax.
That matters because Moderna's medium-term strategy is built around respiratory scale and lifecycle management:
When IP risk is removed, commercial partners, payers, and procurement teams tend to treat supply and pricing as less legally fragile. That does not guarantee demand, but it reduces one category of "unforced error."
What Risks Remain After the Settlement?
This settlement is important, but it does not turn Moderna into a low-risk stock. It simply changes which risks dominate.
1) Pipeline and Regulatory Execution Risk Still Dominates the Multiple
Moderna needs to secure approvals and successfully launch products to restore its recurring revenue after COVID-19, even if the IP overhang decreases.
Investors continue to evaluate Moderna based on timing, trial design, and regulatory alignment across its respiratory and oncology programs.
2) Litigation Risk Is Reduced, Not Erased
The settlement text itself notes that Pfizer and BioNTech litigation related to Genevant and Arbutus patents is ongoing, which highlights that the broader "who owns what" in mRNA delivery and manufacturing remains a live issue in the industry.
3) The Appeal-Triggered Payment Remains a Real Tail Risk
Investors must still consider the possibility that the all-in bill could become significantly larger until the Section 1498 issue is resolved.
Final Takeaway for Traders
If you want a realistic view of Moderna in 2026, it helps to separate three "buckets" of value:
Core cash and base business: what the market thinks the existing commercial portfolio can generate.
Pipeline upside: the probability-weighted value of respiratory, oncology, and other programs.
Risk discounts: legal, regulatory, and execution uncertainty.
This settlement mainly reduces the third bucket, but it also consumes cash, which can slightly pressure the first bucket. The second bucket still has to be earned through data and approvals.
Frequently Asked Questions
What Is Moderna's $950M Settlement About?
Moderna agreed to pay $950 million to settle patent litigation brought by Genevant Sciences and Arbutus Biopharma over lipid nanoparticle delivery technology used in its COVID-19 vaccines.
Could Moderna End Up Paying More Than $950M?
Yes. The settlement includes a potential additional payment of up to $1.3 billion tied to an appellate ruling involving Moderna's Section 1498 defense.
Does This Settlement Change Moderna's Long-Term Outlook?
It can help by removing a major legal overhang and allowing investors to focus more on execution. However, Moderna's long-term value still depends on commercial growth beyond COVID and progress across its pipeline over time.
Conclusion
In conclusion, Moderna's $950 million settlement is a meaningful de-risking event because it clears a long-running LNP patent dispute and removes a legal cloud over parts of Moderna's respiratory vaccine portfolio.
The market's positive reaction makes sense because certainty is valuable, especially when future launches and pricing could have been constrained by litigation.
At the same time, the decision is not a free win. The $950 million payment is a material cash outflow scheduled for Q3 2026, and the additional contingent $1.3 billion tied to the Section 1498 appeal keeps a meaningful tail risk alive.
Disclaimer: This material is for general information purposes only and is not intended as (and should not be considered to be) financial, investment or other advice on which reliance should be placed. No opinion given in the material constitutes a recommendation by EBC or the author that any particular investment, security, transaction or investment strategy is suitable for any specific person.




























