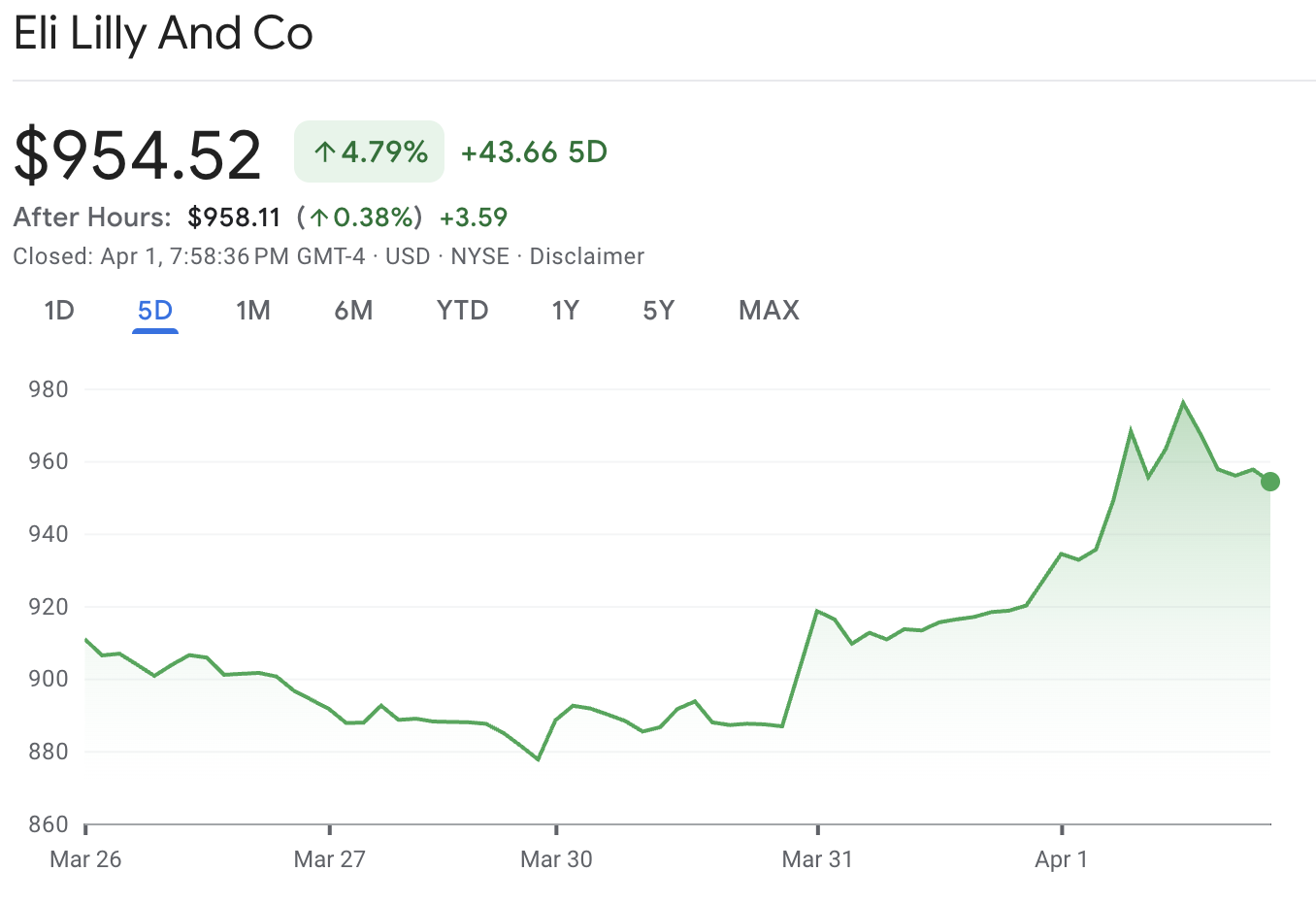
LLY stock rose 3.8% to $954.52 on April 1 after the FDA approved Foundayo, Eli Lilly’s new oral obesity pill.
Even after that jump, the shares were still about 19% below their January peak, which is why investors are asking a bigger question than whether the news was positive.
Now, the question is whether an easier-to-use oral option can expand the addressable market, support Lilly’s 2026 revenue guidance of $80 billion to $83 billion, and sharpen its competitive position against Novo Nordisk.

How LLY Stock Reacted to the Foundayo Approval
LLY closed at $954.52 on April 1, up $34.75 or 3.78% on the session after the FDA approval. Intraday, the stock traded as high as $976.68, suggesting investors viewed the approval as a meaningful catalyst rather than routine regulatory news.
The move also stood out against the broader market. The S&P 500 rose 1.2% on the day, while Lilly outperformed that gain, suggesting the stock-specific reaction was stronger than the broader market tailwind.
Lilly shares also remain about 19% below their January peak, so the approval helped sentiment but did not fully erase the earlier pullback.
What Foundayo Means for LLY Stock
Foundayo gives Lilly a second commercial format in obesity, which matters because oral access can widen the treated population without relying solely on injectable growth.
The FDA approved the pill on April 1, and Lilly plans to start shipping through LillyDirect on April 6, providing investors with a near-term commercial catalyst rather than a distant pipeline story.
The launch also comes with a clear pricing strategy. Lilly said eligible commercially insured patients may pay as little as $25 a month, self-pay pricing starts at $149, and some Medicare beneficiaries may be able to access the drug for $50 a month under the company’s prior access agreement with the U.S. government.
Current Performance of Eli Lilly (LLY) Stock
| Period |
End Price |
Start Price |
Growth |
| 1D |
$954.52 |
$919.77 |
+$34.75 (+3.78%) |
| 1W |
$954.52 |
$916.31 |
+$38.21 (+4.17%) |
| 6M |
$954.52 |
~$825.42 |
~+$129.10 (+15.64%) |
The six-month gain shows the stock was already in an established uptrend before the approval, while the one-day move reflects catalyst-driven repricing rather than a reversal from a broken trend.
The Competitive Landscape: Lilly vs Novo
Novo Nordisk reached the U.S. market first with oral Wegovy in December 2025, but Lilly’s stock case is more about economics than timing. Foundayo gives Eli Lilly an oral entry into obesity with a product that could help expand the market rather than split existing demand.
For investors, Lilly’s advantage is scale, since Foundayo is a small-molecule drug that should be easier to manufacture than Novo’s peptide-based pill.
Lilly is also seeking approval in more than 40 countries and says its U.S. manufacturing commitments since 2020 now exceed $50 billion. That supports the bullish case for stronger supply, better margin control, and wider global revenue capture.
Foundayo Pricing and Access
Foundayo will launch with tiered pricing, with self-pay costs starting at $149 a month and rising by dose. Lilly said eligible commercially insured patients may pay as little as $25 a month, while some Medicare beneficiaries may be able to access the drug for $50 a month from July 1.
For insured patients, the picture is more accessible:
Commercial insurance: as low as $25/month with a Lilly savings card
Self-pay: $149 to $349/month, depending on dose
Medicare Part D: $50/month starting July 1, 2026
LillyDirect is accepting prescriptions immediately, with shipping set to begin on April 6.
What’s Next for LLY Stock
Foundayo adds a fresh catalyst, but the bigger LLY story is still earnings durability and pipeline depth.
The approval supports near-term sentiment because it broadens Lilly’s obesity franchise, but investors are still paying for what comes after this launch and whether the company can keep extending growth beyond its current blockbusters.
Retatrutide: This remains the higher-upside pipeline asset. If later-stage data confirms stronger efficacy, Lilly could deepen its lead in obesity and create another major revenue engine above Zepbound.
Centessa acquisition: The $7.8 billion deal matters because it gives Lilly another growth lane outside metabolic health. That helps diversify the long-term revenue mix and supports a broader valuation case.
Track record: Lilly’s five-year share performance explains why the market keeps rewarding new catalysts. Foundayo does not change that on its own, but it reinforces the view that Lilly is still executing against a strong product cycle.
Risk: Investors will need to see uptake, pricing traction, and follow-through from management before treating this as a lasting re-rating.
What Could Limit the Upside for LLY Stock
The Eli Lilly stock still has to prove that launch demand is broad, reimbursement is workable, and early discontinuation does not reduce the commercial payoff.
There is also a market reality here: LLY bounced on the news, but the shares had already pulled back from their January high.
That leaves investors asking whether Lilly can sustain its premium valuation as competition deepens and the obesity trade matures.
Frequently Asked Questions (FAQ)
1) Why did LLY stock rise after the approval?
The approval adds a second obesity medicine to Lilly’s portfolio and opens a large oral market opportunity. Investors are reacting to the combination of convenience, pricing, and near-term launch readiness.
2) What is the next catalyst for Eli Lilly stock?
The April 30 earnings call is the next key checkpoint. Investors will be looking for management commentary on Foundayo demand, access, launch execution, and the broader obesity pipeline.
3) How does Foundayo change the Lilly vs Novo investment case?
Foundayo gives Lilly an oral obesity product that can compete more directly with Novo while broadening the market beyond injectables. For investors, the key difference is commercial scale, pricing flexibility, and Lilly’s ability to turn convenience into faster revenue growth.
4) What does Lilly need to prove after the Foundayo launch?
Lilly needs to show strong early demand, broad reimbursement, and steady refill rates. Investors will also want proof that Foundayo can add meaningful sales without weakening the company’s premium valuation or slowing momentum in its wider obesity franchise.
Summary
Foundayo matters because it gives Eli Lilly a new commercial growth lever, not just a short-term headline catalyst. It gives the company a new way to expand its obesity franchise, bring more patients into treatment, and strengthen its position against Novo Nordisk in the fast-growing oral GLP-1 market.
For LLY stock, the real story now is execution. If launch demand is strong and access holds up, Foundayo can support Lilly’s next phase of revenue growth rather than simply trigger a short-term rally.
Healthcare stocks tend to command premium valuations when they combine pricing power, pipeline depth, and visible earnings growth, and Lilly remains one of the clearest examples of that setup.
Disclaimer: This material is for general information purposes only and is not intended as (and should not be considered to be) financial, investment or other advice on which reliance should be placed. No opinion given in the material constitutes a recommendation by EBC or the author that any particular investment, security, transaction or investment strategy is suitable for any specific person.
Sources
FDA Approval of Foundayo
Eli Lilly Q4 2025 Earnings and 2026 Guidance
Eli Lilly Retatrutide
Eli Lilly Plans $5 Billion Manufacturing Facility in Virginia