Key Takeaways
Revolution Medicines shares surged after Phase 3 data showed its pancreatic cancer drug candidate extended survival compared to chemotherapy.
The company reported a median overall survival of 13.2 months with daraxonrasib, versus 6.7 months with standard treatment.
The trial met all primary and key secondary endpoints, supporting future regulatory filings.
Broader biotech shares also rose, though RVMD’s gain significantly outpaced the sector.
The rally improved sentiment for clinical-stage biotech, but the sector remains focused on selective, data-driven winners.
Revolution Medicines (RVMD) stock rose after pivotal pancreatic cancer data brought the company back to investors’ attention. Shares jumped following late-stage trial results showing a clear survival benefit for daraxonrasib in previously treated metastatic pancreatic cancer. RVMD closed at $136.30, up $39.84, or about 41.3%.
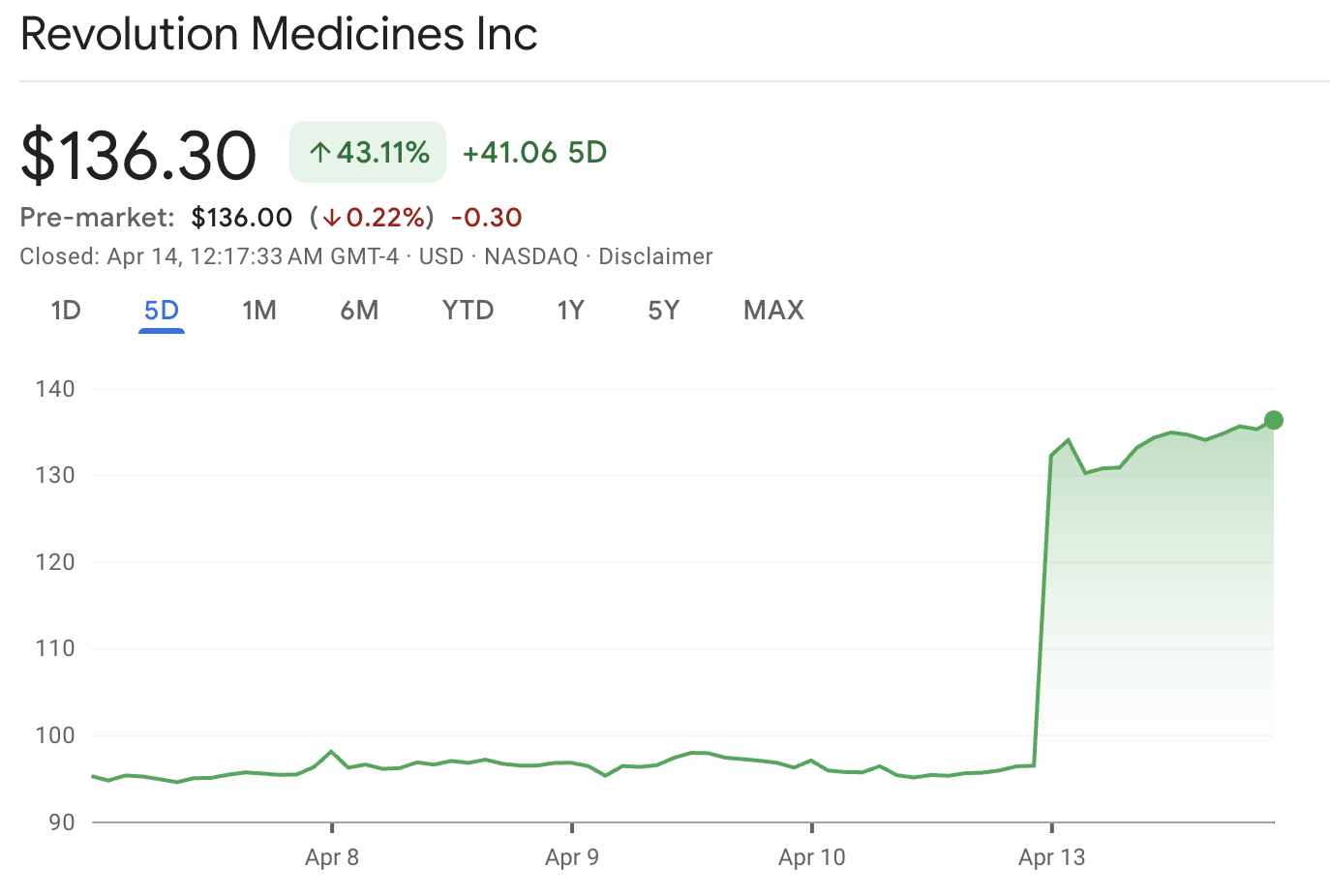
This move stood out because it was driven by a clear clinical catalyst in a challenging oncology area. It also came at a time when investors have favored only biotech companies with strong evidence and a clear regulatory path.
What Drove The RVMD Stock Higher
Revolution Medicines reported that daraxonrasib achieved statistically significant and clinically meaningful improvements in both progression-free and overall survival in the Phase 3 RASolute 302 study. The trial met all primary and key secondary endpoints.
The key result was overall survival: in the intent-to-treat population, median overall survival was 13.2 months for daraxonrasib compared to 6.7 months for standard chemotherapy, with a hazard ratio of 0.40.
This result is significant because metastatic pancreatic ductal adenocarcinoma is among the deadliest cancers. Revolution Medicines noted that pancreatic cancer affects about 60,000 people annually in the United States and causes approximately 50,000 deaths, with a five-year survival rate for metastatic disease of about 3%.
Why The Data Carried Weight
This was not an early-stage or small subgroup update. RASolute 302 is a global, randomized Phase 3 registrational trial in previously treated metastatic pancreatic cancer, with an enrollment of 501 patients.
The treatment arm used 300 mg once-daily oral daraxonrasib, while the control arm received the investigator’s choice of standard chemotherapy. Revolution Medicines also said the drug was generally well-tolerated and showed no new safety signals in the interim analysis.
This combination of efficacy and tolerability gave investors confidence that the result could have regulatory and commercial significance, not just scientific value.
What Revolution Medicines Stock Surge Means for Biotech
Current Biotech Industry Performance
Biotech is trading better than the broader healthcare complex, but its strength remains selective rather than broad-based. In the latest U.S. session, the SPDR S&P Biotech ETF (XBI) rose to $132.05, up about 2.0%, while the iShares Biotechnology ETF (IBB) gained to $172.19, up about 1.6%.
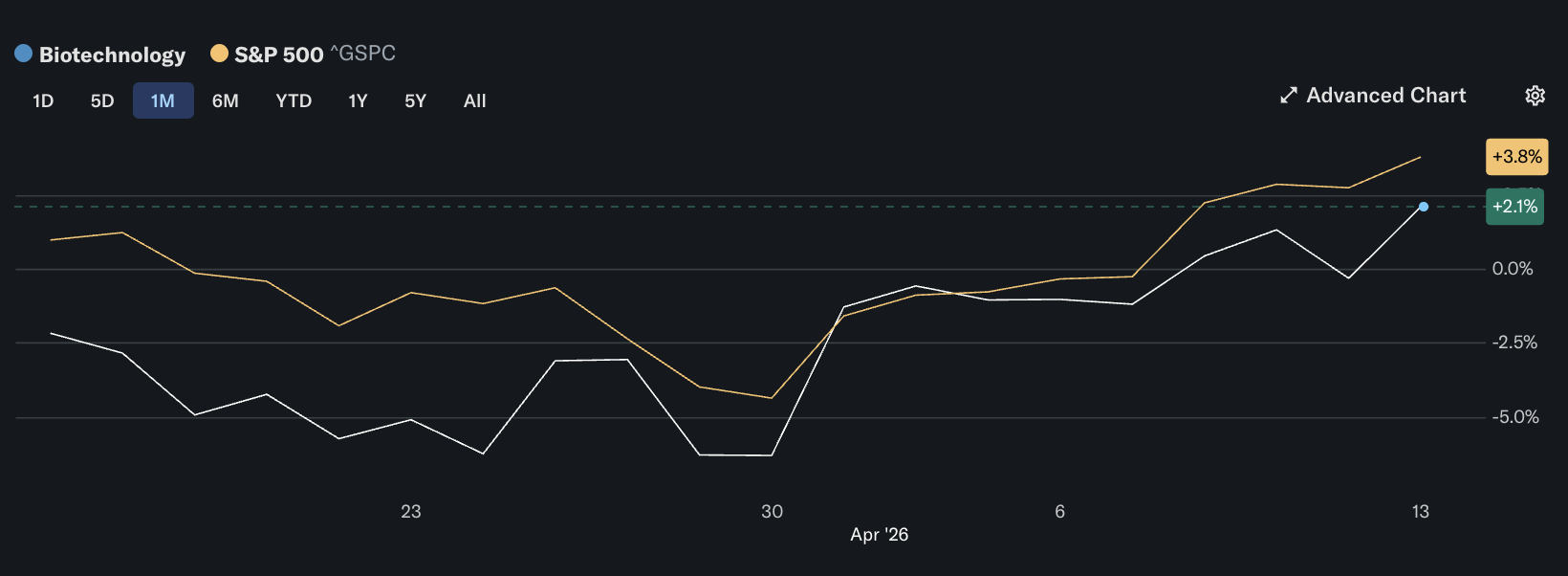
By comparison, the Health Care Select Sector SPDR ETF (XLV) was up a more modest 0.45% at $147.97. Biotech outperformed the broader healthcare sector, but the gains remained concentrated in a small group of catalyst-driven names.
A few points stand out from the current tape:
Leadership is coming from catalyst-driven names. An industry report shows Revolution Medicines up 41.35%, Spyre Therapeutics up 23.36%, Cellectis up 19.22%, and Ideaya Biosciences up 7.61%, suggesting investors are rewarding clinical and pipeline news rather than indiscriminately buying the whole group.
The move is stronger than healthcare, but narrower than a true rotation. XBI and IBB are both outperforming XLV, yet the outsized winners are concentrated in companies with fresh data or company-specific headlines. That usually signals selective conviction, not a blanket rush into defensives.
Industry sentiment also has support from deal activity. A Wall Street Journal report said 19 biotech deals above $1 billion have been announced so far in 2026, with none above $10 billion, as larger drugmakers favor smaller acquisitions to refill pipelines ahead of patent expirations. That backdrop can help sustain interest in quality biotech names even when the market is not chasing the entire sector.
Why RVMD Now Carries More Weight
Revolution Medicines is not trading on a single concept. The company stated that daraxonrasib is being evaluated in four global Phase 3 registrational trials: three in pancreatic cancer and one in non-small cell lung cancer.

That provides investors with a broader framework for valuing the company’s pipeline. The latest trial result may strengthen confidence not only in daraxonrasib but also in Revolution Medicines’ broader approach to targeting RAS-driven cancers.
The Balance Sheet Helps The Story
Another factor supporting the move is the company’s capital strength. Revolution Medicines reported $2.0 billion in cash, cash equivalents, and marketable securities at the end of 2025.
While this does not eliminate execution risk, it reduces near-term financing pressure, which is important for a late-stage oncology company preparing for additional trials and regulatory filings.
Revolution Medicines also guided for $1.6 billion to $1.7 billion in 2026 GAAP operating expenses, reflecting the scale of its development program.
The Caution Investors Should Keep in Mind
The rally was based on solid data, but the story is ongoing. Daraxonrasib remains investigational and is not approved by regulators in the United States or Europe.
The company intends to include these results in a future New Drug Application to the FDA and other regulators. It also plans to present detailed data at the 2026 American Society of Clinical Oncology Annual Meeting.
That leaves three near-term issues for investors:
whether fuller data hold up under broader clinical scrutiny
how quickly regulators move
whether the company can convert clinical success into a durable commercial franchise
What The Market May Watch Next
For now, the stock’s move suggests investors are willing to revisit biotech when a catalyst is significant enough to shift expectations.
The move does not yet amount to a broad biotech rotation. It does, however, show that investors are willing to reward late-stage clinical winners again.
Revolution Medicines is now an important test case. If the company delivers detailed data, steady regulatory progress, and continued pipeline execution, RVMD’s move could become more than a one-day event.
Frequently Asked Questions (FAQ)
Why did Revolution Medicines stock rise?
The stock rose after the company reported Phase 3 data showing that daraxonrasib improved survival in previously treated metastatic pancreatic cancer compared with chemotherapy.
How much did RVMD gain?
RVMD closed at $136.30, up $39.84, which is roughly a 41.3% gain on the session.
What is daraxonrasib?
Daraxonrasib is an investigational oral RAS(ON) inhibitor being developed for cancers driven by a broad range of RAS mutations.
Does this mean biotech is broadly back?
Not yet. The session showed selective demand for strong clinical stories, but broader biotech ETFs rose far less than RVMD.
Summary
Revolution Medicines’ rally was driven by a result that the market could clearly measure. A late-stage pancreatic cancer study showed that daraxonrasib extended survival compared with chemotherapy, and investors responded by sharply repricing the stock.
The wider message is narrower than the stock move itself. Biotech appears to be back in play for companies that can deliver decisive clinical data, but the sector has not turned into a uniform risk-on trade.
For now, RVMD looks more like a stock-specific repricing than proof of a full healthcare shift.
Disclaimer: This material is for general information purposes only and is not intended as (and should not be considered to be) financial, investment or other advice on which reliance should be placed. No opinion given in the material constitutes a recommendation by EBC or the author that any particular investment, security, transaction or investment strategy is suitable for any specific person.
Sources
Revolution Medicines press release: Daraxonrasib Demonstrates Overall Survival Benefit in Phase 3 RASolute 302 Trial
Revolution Medicines investor update: Fourth Quarter and Full Year 2025 Financial Results and Update on Corporate Progress
ClinicalTrials.gov: RASolute 302 Study Details




























